Measuring and comparing it with the number for an atom, he discovered that the mass of the electron was very small, merely 1/1,836 that of a hydrogen ion. This ratio was known for atoms from electrochemical studies. Accordingly, he called his particles electrons.įrom the magnitude of the electrical and magnetic deflections, Thomson could calculate the ratio of mass to charge for the electrons. Thomson’s discovery established the particulate nature of electricity. 
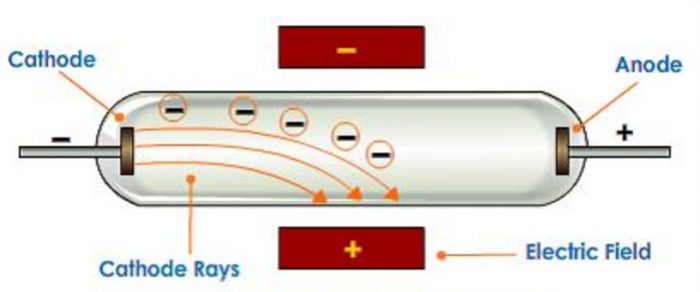
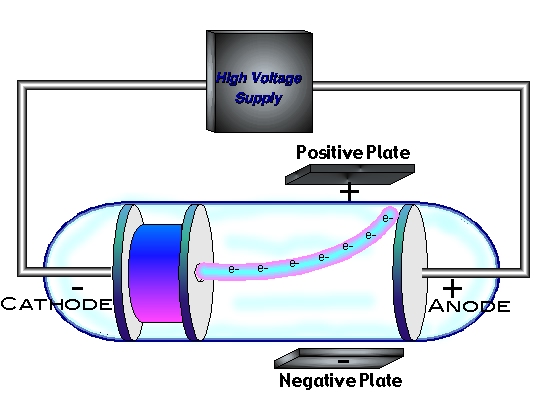
With both magnetic and electric deflections observed, it was clear that cathode rays were negatively charged particles. The deflection was proportional to the difference in potential between the plates. When the top aluminum plate was negative, the rays moved down when the upper plate was positive, the rays moved up. He directed the cathode rays between two parallel aluminum plates to the end of a tube where they were observed as luminescence on the glass. Thomson repeated Hertz’s experiment with a better vacuum in 1897. Thomson thought Hertz’s vacuum might have been faulty and that residual gas might have reduced the effect of the electric field on the cathode rays. Heinrich Hertz, the aforementioned German physicist, reported that the cathode rays were not deflected when they passed between two oppositely charged plates in an 1892 experiment. A crucial test of the nature of the cathode rays was how they would be affected by electric fields. Most German physicists, on the other hand, believed that the rays were waves because they traveled in straight lines and were unaffected by gravity. Most French and British physicists, influenced by Crookes, thought that cathode rays were electrically charged particles because they were affected by magnets. By the late 1880s the controversy over the nature of cathode rays had divided the physics community into two camps. As a result of Crookes’s work, cathode rays were widely studied, and the tubes came to be called Crookes tubes.Īlthough Crookes believed that the particles were electrified charged particles, his work did not settle the issue of whether cathode rays were particles or radiation similar to light. As the luminescence did not depend on what gas had been in the vacuum or what metal the electrodes were made of, he surmised that the rays were a property of the electric current itself. English physicist and chemist William Crookes investigated cathode rays in 1879 and found that they were bent by a magnetic field the direction of deflection suggested that they were negatively charged particles. The shadow proved that the cathode rays originated from the cathode. Hittorf saw a shadow cast by an object placed in front of the cathode. In 1869, with better vacuums, Plücker’s pupil Johann W. He found a green glow on the wall of his glass tube and attributed it to rays emanating from the cathode. Plücker discovered cathode rays in 1858 by sealing two electrodes inside the tube, evacuating the air, and forcing electric current between the electrodes. The existence of the electron showed that the 2,000-year-old conception of the atom as a homogeneous particle was wrong and that in fact the atom has a complex structure.Ĭathode-ray studies began in 1854 when Heinrich Geissler, a glassblower and technical assistant to German physicist Julius Plücker, improved the vacuum tube. Their work culminated in the discovery by English physicist J.J.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
